INTRODUCTION
Thrombosis is one of the leading causes of morbidity and mortality worldwide and represents a major public health problem. Venous thromboembolism (VTE), which includes deep vein thrombosis and pulmonary embolism is considered the third most common cardiovascular disorder after coronary artery disease and stroke [1]. Its etiology is multifactorial and results from the interaction between genetic and acquired factors that alter the balance of the hemostatic system.
These conditions may be classified as inherited thrombophilias, associated with genetic variants affecting proteins involved in hemostasis, and acquired thrombophilias, related to clinical factors such as surgery, pregnancy, prolonged immobilization, use of hormonal contraceptives, or inflammatory diseases [2,3].
Among the genetic variants most widely studied in relation to the thrombosis risk are Factor V Leiden (G1691A), prothrombin G20210A, and the methylenetetrahydrofolate reductase gene (MTHFR) C677T variant. Factor V Leiden is one of the most common inherited thrombophilias and has been associated with an increased risk of venous thromboembolism. Likewise, the prothrombin G20210A variant has been linked to increased thrombotic risk [4]. The MTHFR C677T variant, associated with hyperhomocysteinemia, has been widely studied in patients with thrombotic events and recurrent pregnancy loss;however, its direct association with venous thrombosis remains controversial in the scientific literature [5].
During pregnancy, a physiological hypercoagulable state develops, increasing thrombotic risk. This phenomenon has also been linked to obstetric complications such as recurrent pregnancy loss (RPL), defined as the loss of two or more consecutive pregnancies before 20 weeks of gestation (American College of Obstetricians and Gynecologists [ACOG]) [6]. Although the etiology of RPL is multifactorial, inherited thrombophilias have been investigated as possible contributing factors in a subgroup of patients.
In Latin America, and particularly in Panama, information on the prevalence of genetic variants associated with thrombophilia is limited. Therefore, this study aimed to determine the prevalence of Factor V Leiden (G1691A), prothrombin G20210A, and MTHFR C677T variants in patients with thromboembolic events and recurrent pregnancy loss, as well as in healthy controls from a Panamanian population.
MATERIALS AND METHODS
Study design
An observational, analytical, population-based case-control study was conducted to determine the prevalence of Factor V Leiden (G1691A), prothrombin G20210A, and MTHFR C677T genetic variants in a Panamanian population.
Study population
The study population consisted of patients evaluated at the Genetics Laboratory of Dr. Arnulfo Arias Madrid Hospital Complex, Panama.
Two study groups were included:
Group A (recurrent pregnancy loss): Fifty-five Panamanian women with a history of two or more recurrent pregnancy loss, referred for genetic thrombophilia testing since January 2013, were included. The control group consisted of 55 women without a history of recurrent pregnancy loss or thrombosis and with at least one term pregnancy.
Group B (thromboembolic events)
One hundred five patients with a history of thromboembolic events, referred by the Prenatal Diagnosis, Hematology, Gynecology-Obstetrics, and Neurology departments, were included. These patients had experienced stroke, deep vein thrombosis, or pulmonary thromboembolism without other associated risk factors. The corresponding control group consisted of 105 Panamanian individuals without a history of thrombosis.
Participants’ ages ranged from 18 to 44 years in the RPL group and from 20 to 70 years in the thromboembolic event group.
Patients with primary or acquired immunosuppression, acute infectious processes, cancer, or any condition that impaired understanding of the study, whether due to neurological or psychiatric disease or language barriers, were excluded.
Sample collection and DNA extraction
Peripheral blood collected in EDTA anticoagulant tubes was used for DNA extraction. Genomic DNA was extracted from 200 μL of blood using the QIAamp DNA Mini Blood kit and the automated QIAcube system (QIAGEN), following the manufacturer’s instructions [7].
Detection of genetic variants
Identification of Factor V Leiden (G1691A), prothrombin G20210A, and MTHFR C677T variants was performed by endpoint multiplex PCR using the Elucigene TRP thrombophilia kit (Gen-Probe). The method is based on the ARMS-PCR (Amplification Refractory Mutation System) approach [8], which allows detection of point mutations through allele-specific amplification [9], following the manufacturer’s instructions. Each reaction included internal amplification controls. Amplified products were separated by electrophoresis on a 2% agarose gel and visualized under ultraviolet light to determine the corresponding genotype.
Statistical analysis
Genotypic and allelic frequencies of the studied variants were calculated. Continuous variables, such as age, were described using the mean and range. The association between genetic variants and thrombotic events or recurrent pregnancy loss was evaluated using 2×2 contingency tables and calculation of odds ratios (OR) with 95% confidence intervals (95% CI) [10].
RESULTS
A total of 320 individuals were analyzed, distributed into two study groups and their corresponding controls. Group A included 55 women with recurrent pregnancy loss (RPL), aged 18 to 44 years (mean age: 32 years), and 55 controls with at least one term pregnancy and no history of RPL or thrombosis. Group B consisted of 105 patients with thromboembolic events (TEE), aged 20 to 67 years (mean age: 37 years), of whom 90% were women and 10% were men. The corresponding control group included 105 healthy individuals, aged 20 to 75 years (mean age: 42 years), of whom 84% were women and 16% were men.
Factor V Leiden (G1691A) and prothrombin G20210A variants were not detected in patients with recurrent pregnancy loss (Table 1). Only one heterozygous individual for prothrombin G20210A was identified in the control group, corresponding to a prevalence of 0.9%. Regarding the MTHFR C677T variant, 22 heterozygous patients (40%), and 16 homozygous patients (29.1%) were observed in the RPL group, whereas 26 heterozygous controls (47.3%) and 12 homozygous controls (21.8%) were identified in the control group. Allelic prevalence was 49.1% in patients and 45.5% in controls, with no evidence of association with recurrent pregnancy loss (OR = 1.0;95% CI: 0.44–2.24).

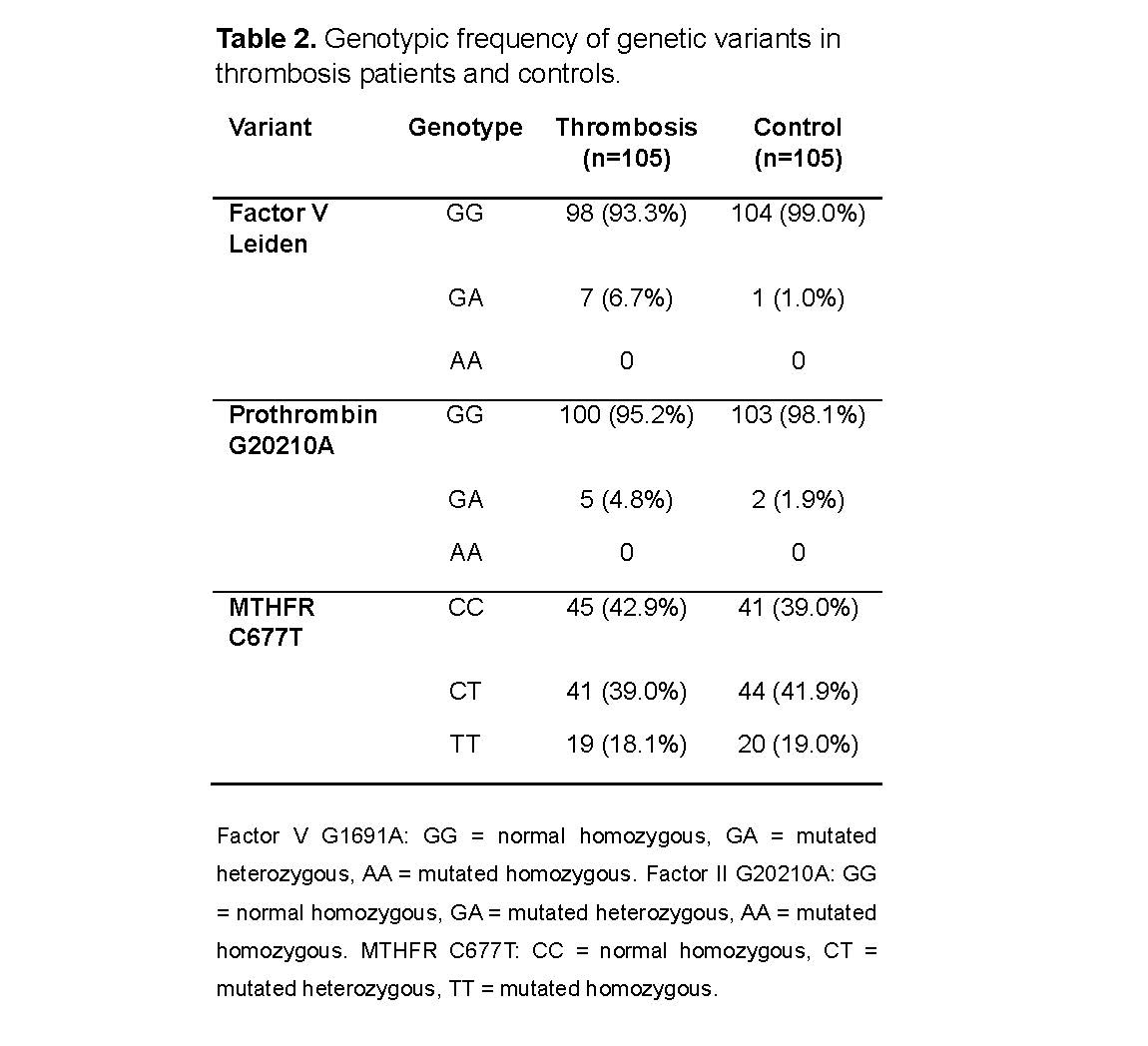
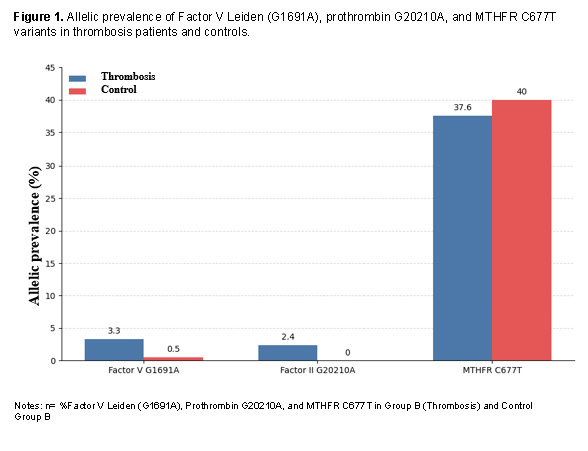
In the thrombosis group, 7 heterozygous individuals for Factor V Leiden (6.7%) were identified (Table 2), whereas 1 heterozygous individual (1.0%) was detected in the control group, with no homozygous cases. This variant showed an association with increased thrombosis risk (OR = 7.43;95% CI: 0.89–61.48). The prothrombin G20210A variant was detected in 5 thrombosis patients (4.8%) and 2 controls (1.9%), all heterozygous, with a moderate association with thrombosis (OR = 2.58;95% CI: 0.48–13.58). Regarding the MTHFR C677T variant, 41 heterozygous patients (39.0%) and 19 homozygous patients (18.1%) were observed in the thrombosis group, whereas 44 heterozygous controls (41.9%) and 20 homozygous controls (19.0%) were identified in the control group. Allelic prevalence was 37.6% in patients and 40.0% in controls, with no evidence of association with thromboembolic events (OR = 0.85;95% CI: 0.49–1.48) (Figure 1).
DISCUSSION
The results of the present study indicate that the Factor V Leiden (G1691A) variant is associated with an increased risk of thrombosis in the analyzed population. Heterozygosity of 6.7% was observed in patients with thromboembolic events, compared with 1.0% in controls, corresponding to an approximately sevenfold increase in thrombosis risk (OR = 7.43;95% CI: 0.89–61.48). This finding is consistent with previous studies showing a significant increase in thromboembolic risk among heterozygous carriers of this variant, which leads to resistance to activated protein C and promotes a prothrombotic state [11]. When compared with results reported in other populations, variable heterozygosity frequencies are observed, including 2.6% in Argentina, 5.4% in Chile, 6% in Brazil, and 8% in the United States, while prevalences of 28% in Costa Rica and 14.3% in Nicaragua have been reported in Central America [12,13,14,15,16,17]. These differences reflect the genetic heterogeneity among populations;however, the frequency observed in the Panamanian population falls within the range reported in the region.
Regarding the prothrombin G20210A variant, a prevalence of 2.4% was observed in thrombosis patients and 1.0% in controls, with heterozygous frequencies of 4.8% and 1.9%, respectively, suggesting a moderate association with thrombotic risk (OR = 2.58;95% CI: 0.48–13.58), lower than that observed for Factor V Leiden. The frequency found is comparable to that reported in Chile (5.4%), the United States (5.0%), and Argentina (5.1%), and slightly higher than that reported in Brazil (3.0%) [13,15,12,14].
The relationship between inherited thrombophilias and recurrent pregnancy loss (RPL) remains controversial. Although some studies have suggested a possible association, the available evidence is heterogeneous and routine screening in women without adverse obstetric history is not widely recommended [18]. In agreement with this context, Factor V Leiden and prothrombin G20210A variants were not detected in the RPL group analyzed in this study, and therefore no association with this condition was observed. This result is consistent with studies conducted in Latin American populations, such as a cohort study in Argentina that also found no significant differences in the distribution of thrombophilic variants between patients with recurrent pregnancy loss and fertile controls [19].
As for the MTHFR C677T variant, similar frequencies were observed between patients and controls in both the thrombosis and RPL groups, suggesting that this variant does not constitute an independent risk factor for these conditions in the studied population. Although it may be associated with elevated homocysteine levels, its clinical impact as a thrombotic risk factor is limited, and its high frequency in the general population may explain the absence of a significant association [20,21]. Taken together, these findings support the notion that inherited thrombophilias act mainly as predisposing factors, whose clinical effect depends on interaction with other genetic and environmental factors.
CONCLUSIONS
This study characterized the prevalence of Factor V Leiden, prothrombin G20210A, and the MTHFR C677T variant in Panamanian patients with thromboembolic events and recurrent pregnancy loss (RPL). A strong association was observed between Factor V Leiden and thrombosis, with an approximately sevenfold increase in thrombotic risk, whereas prothrombin G20210A showed a moderate association. No association was found between these variants and RPL, nor between the MTHFR C677T variant and thrombotic or gestational events. These findings highlight the importance of inherited thrombophilia testing in patients with thrombosis to support clinical decision-making and family risk assessment.
Limitations and recommendations
Prospective studies are recommended to evaluate folic acid and vitamin B12 concentrations, both cofactors of the MTHFR enzyme, in patients with the homozygous genotype, and to correlate them with plasma homocysteine levels, given that hyperhomocysteinemia has been associated with increased thrombotic risk. It would also be relevant to explore the possible genetic interaction between Factor V Leiden and MTHFR variant, as well as their coexistence with other inherited thrombophilias, in order to evaluate their synergistic effect on the predisposition to thrombotic events.
Acknowledgments
To the Genetics Laboratory of Dr. Arnulfo Arias Madrid Hospital Complex, Social Security Fund, Panama, for its support in carrying out the laboratory determinations.
Author contributions
JC: test standardization, conceptualization, data analysis, manuscript drafting, revision, and writing. OE: manuscript conceptualization. LDG: conceptualization and manuscript writing support. LS: review, protocol correction, and protocol writing.
Conflict of interest / Disclosure
All authors declare that they participated in the design, execution, and analysis of the article and approved the final version. In addition, there are no conflicts of interest related to this article.
Data availability
All data supporting the findings of this study are available within the article. Additional information is available from the corresponding author upon reasonable request.
