Introduction
Proteus syndrome is a rare syndrome characterized by disproportionate growth of the limbs, multiple hamartomas, and vascular malformations. Cerebriform connective tissue nevi, also called cerebriform plantar hyperplasia, are present in most patients and are the main feature of the syndrome. If present, even in isolation, they can be considered a pathognomonic sign. The syndrome could be associated with mosaicism with a somatic activating mutation in the AKT1 gene, located on chromosome 14q32.3. Several allelic mutations occur at the gene locus responsible for Proteus syndrome, which triggers the overgrowth of somatic tissue.
This syndrome is poorly described and its estimated prevalence ranges from 1:1,000,000 to 1:10,000,000 inhabitants. Fewer than 100 cases have been described in the literature (Biesecker, et. al, 2020). Its rare occurrence justifies this report (Soriano et al., 2017). This literature review presents several confirmed cases of the disease.
Description of the PI3K/AKT 1 pathway in patients with Proteus syndrome
Proteus syndrome is related to a dysregulation of the PI3K-AKT pathway. On one hand, the PI3K protein is phosphorylated, activating transcriptional activation of the vascular endothelial growth factor (VEGF) and the expression of the hypoxia-inducible factor 1α (HIF1α) angiogenesis, growth, senescence, and other processes, through molecular mechanisms (See Figure 1 and 2) (Pinzón C, Serrano M, Sanabria M, 2009).
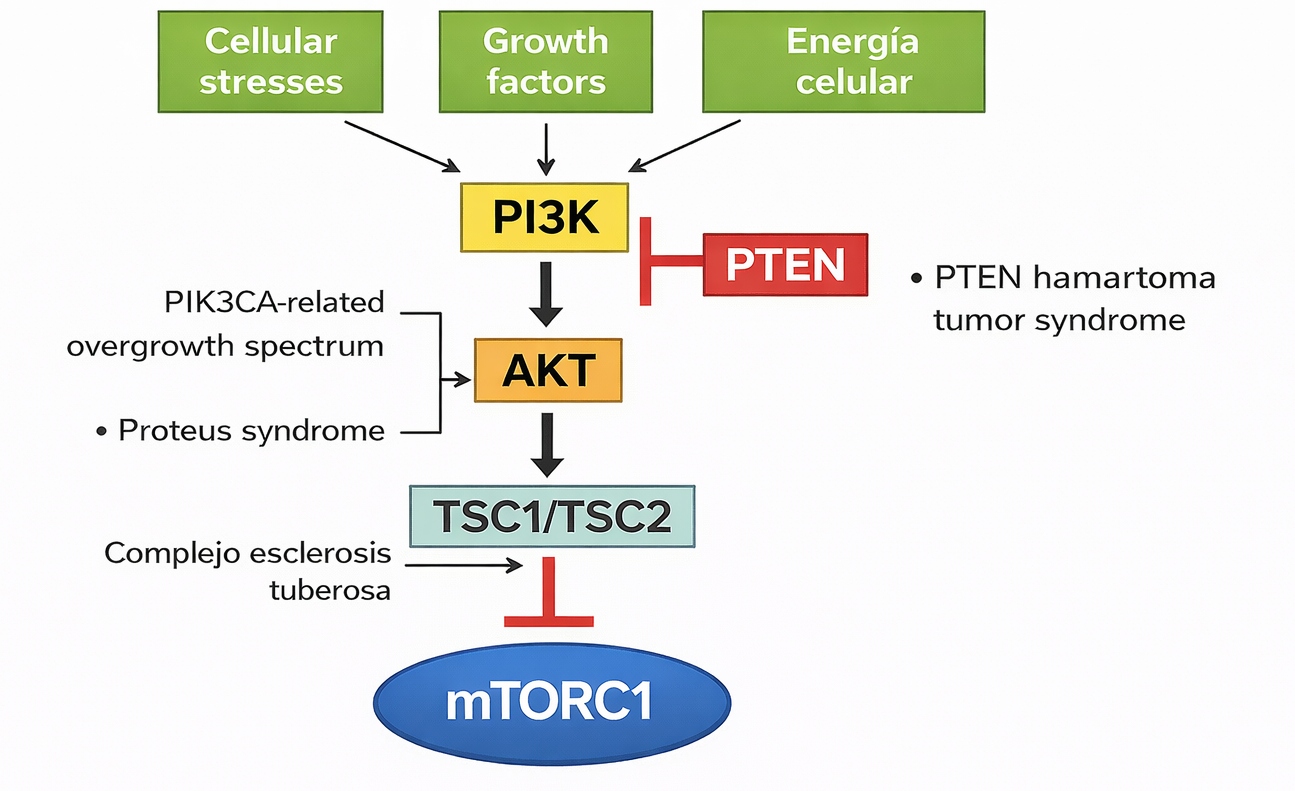
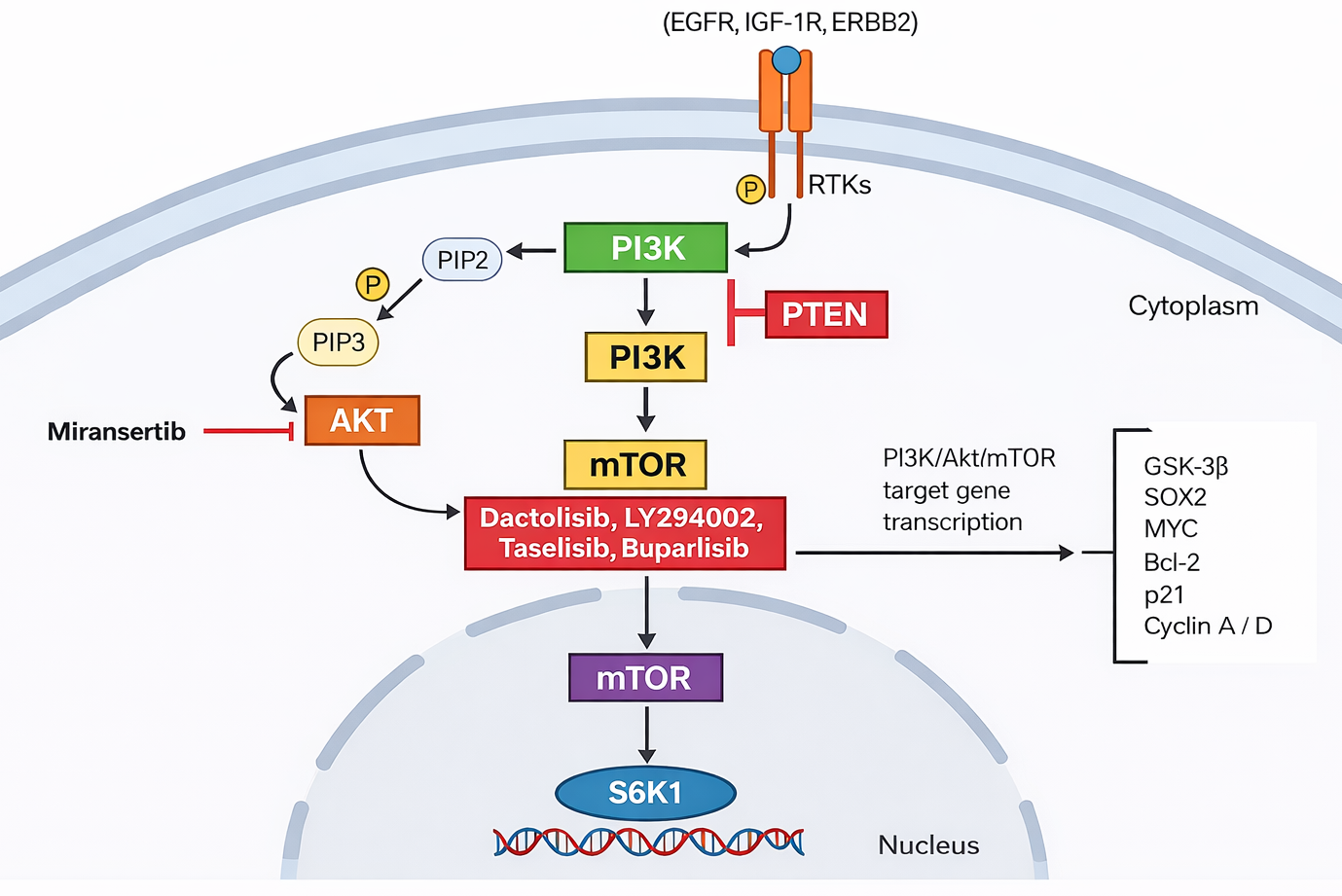
The AKT gene encodes a protein that participates in the regulation of growth, division (proliferation), and death of cells. This gene encodes the AKT protein that phosphorylates cyclin-dependent kinase inhibitors (CKI) p21CIP1/WAF1 and p27KIP1, which translocate to the cytoplasm and are degraded. CKI proteins have an antiproliferative effect, therefore when degraded, cell proliferation is deregulated as it increases. Additionally, the AKT protein inhibits glycogen synthase kinase-3 (GSK3) by phosphorylation, affecting glucose metabolism.
In patients with Proteus syndrome who present a mutation in AKT1 in the form of somatic mosaicism (c. 49G>A, p.Glu17Lys), this causes AKT or PDK to be always active, thus affecting the regulation of processes in the body as there is an overproduction of transcription factors and increased cell growth rates, so cells continue to divide without dying, involved. The increase in cell proliferation in some tissues and organs results in the characteristics of the syndrome such as abnormal growth and increased risk of developing tumors (National Institutes of Health, 2017)
Description of the mechanism of action of miransertib on the PI3K/AKT pathway
The drug miransertib binds to the allosteric site of the AKT protein, interfering with its activation and preventing its phosphorylation, thus reducing signaling in the rest of the PI3K/AKT pathway cascade. This drug will inhibit both the active form of AKT and its inactive form, thus stopping its activation and reducing the phosphorylation of proteins involved in cell growth in patients with Proteus syndrome.
Similarly, this blockade of AKT will favor apoptosis and prevent positive feedback of PI3K.
In the case of Proteus syndrome, this decrease in activity showed efficacy in reducing the progression of excessive growth and improving symptoms.
Description of treatment with miransertib in patients with Proteus syndrome
Case 1
In the first clinical trial, a daily dose of 5 mg was applied to a group composed of six patients: three adults and three children, over the course of a year. Throughout this period, a reduction in the size of the lesions was observed, highlighting that in two of the patients a significant decrease in the size of the cerebriform connective tissue was recorded. Additionally, stabilization in the excessive growth of tissues was evidenced in all treated patients, suggesting a positive response to the treatment in terms of controlling the development of the lesions. This was determined by qualitative assessment and through visual evaluations, showing the differences between the different growth rates of CCTN.
Case 2
In the second trial, a 20-year-old male patient was treated. After the initial evaluation, an open-label treatment with miransertib was started, administering a daily oral dose of 10 mg (approximately 5 mg/m²/day). This dose was progressively increased, reaching 30 mg per day (15 mg/m²/day) and, after three months of treatment, adjusted to 50 mg daily (25 mg/m²/day).
After 11 months of treatment, an improvement in the patient's overall well-being was observed according to the IPI. There was greater mobility in the ankle, spine, and hands, as well as a subjective decrease in the excessive growth of the right facial bone. Additionally, a reduction in areas affected by cerebriform connective tissue nevi on the soles of the feet was reported. Whole-body MRI results showed stability, with no evidence of apparent disease progression. There was a total reduction of 14 to 17% in the CCTN growth area.
Case 3
The individual is an 18-year-old man who has participated in the Natural History Study of Proteus Syndrome (Clinicaltrials.gov NCT00001403) since he was 6 years old and enrolled in the pharmacodynamic study when he was 13 years old. When he enrolled in the phase 1 study (Clinicaltrials.gov NCT02594215), he had a history of leg length discrepancy requiring multiple surgeries, bilateral plantar CCTN, musculoskeletal pain in the lower back and lower extremities, eczema, seasonal allergies, iron deficiency anemia, and irritable bowel syndrome. The mosaic variant in AKT1 c.49G > A, p.(Glu17Lys) was present in cultured fibroblasts from an affected skin biopsy.
After completing the 48 weeks of treatment with miransertib originally planned as part of the phase 1 study, the patient expressed a desire to continue treatment due to pain improvement and deceleration of CCTN growth. The protocol was modified to provide continued use of miransertib to evaluate long-term safety and response durability (i.e., continued pain reduction and deceleration of CCTN growth).
There was a 9-month pause before resuming treatment with miransertib at a dose of 5 mg/m2 once a day. Based on the demonstration of safety with daily doses of 15 mg/m2 and 25 mg/m2 from an industry-sponsored expanded access program and a perceived decrease in effect, the dose was increased to 10 mg/m2 per day 16 weeks after resuming treatment with miransertib. The magnitude of the dose escalation, doubling the daily drug exposure, was selected to minimize the potential for adverse events and was based on the dose level of the original 3 + 3 study design of the phase 1 trial. As of the publication date of the article, he continues to take 10 mg/m2 (20 mg per dose) daily.
The individual has bilateral plantar CCTN. The left foot is more severely affected than the right. Before starting miransertib, the left CCTN affected 39.2% of the left plantar surface and the right CCTN affected 9.5% of the right plantar surface. After 48 weeks of treatment, the left CCTN affected 40.3% of the left plantar surface and the right CCTN affected 10.5% of the right plantar surface. Five years after starting treatment with miransertib, the left CCTN affects 44.6% of the left plantar surface and the right CCTN affects 15.2% of the right plantar surface. The annual increase in the percentage of plantar surface affected by CCTN during the 7 years prior to treatment with miransertib was 5.3% and 4.8% on the left and right sides, respectively. The annual increase in the percentage of plantar surface affected by CCTN during the 5 years after starting treatment with miransertib was 1.2% and 1.2% on the left and right sides, respectively.
Case 4
In this case, a 17-year-old female patient began miransertib treatment at age 15. She was diagnosed with Proteus syndrome shortly after birth due to excessive hand growth, resulting in multiple surgical interventions, including finger amputations in both hands, cervical vertebral fusion, and confinement to a wheelchair by age 10.
At age 13, she underwent bilateral salpingo-oophorectomy, hysterectomy, and peritoneal staging for low-grade serous ovarian carcinoma, suggesting a borderline ovarian serous tumor. Twenty months later, pelvic recurrence associated with ascites and partial thrombosis of the extrahepatic portal vein segment was reported.
Due to potential complications, surgical intervention was not pursued despite the clinical significance of portal vein involvement. Additionally, chemotherapy and hormonal therapy with aromatase inhibitors were not recommended due to thrombosis.
Given the lack of effective treatment options, experimental therapy was pursued, with miransertib selected. Prior to treatment, CT imaging revealed a secondary neoplastic pelvic mass (sum of diameters: 52 mm) and multiple peritoneal implants with moderate ascites.
Treatment began at age 15 years and 5 months with an initial dose of 50 mg/day for 5 days followed by 9 days off every two weeks. No side effects were reported during the first 4 weeks.
At week 5, the dose was increased to 100 mg/day for 5 days followed by 5 days off every two weeks.
The first disease evaluation at 2 months showed resolution of ascites and a significant decrease in CA-125 (38 U/mL), with stable measurable disease. CA-125 normalized by week 12.
At week 24, CT imaging demonstrated a partial response (PR) according to RECIST 1.1 criteria, with significant reduction of pelvic mass size (sum of diameters: 39.2 mm), resolution of peritoneal implants, and complete resolution of portal vein thrombosis.
CT scans at 12 and 19 months confirmed ongoing PR with residual pelvic mass volumes of 16.7 mL and 6.6 mL, respectively. At the final follow-up at 22 months, imaging showed complete clinical remission of the secondary lesion.
The patient reported reduced plantar CCTN folds in the left foot, increased range of motion in upper extremities and joints (hands, spine, knees), and these findings were confirmed by clinicians during follow-up visits. She also reported decreased pain in the spine, hands, and feet, and regained the ability to transfer independently from wheelchair to bed and lie supine. Images documented overgrowth lesion changes in hands and feet during treatment.
Discussion and Outcomes
Our study results indicate that miransertib, an AKT inhibitor, reduces the size and growth of certain lesions—particularly those involving excessive tissue overgrowth—and improves mobility in most patients. Notably, decreased CCTN growth and reduction of typical cutaneous manifestations, such as atypical plantar folds, were observed. Benefits were also reported in pain reduction and stabilization of pulmonary and skeletal conditions.
The data suggest stabilization of abnormal growth progression, highlighting the impact of AKT inhibition on the PI3K/AKT pathway by blocking the atypical activity that characterizes Proteus syndrome.
Comparison with previous research supports the effect of AKT inhibitors. Earlier studies demonstrated that miransertib can slow lesion growth, consistent with findings from clinical trials.
Mild side effects were reported, including fatigue, nausea, and headache. Adverse events included dry mouth and one episode of gingivostomatitis.
Overall, these findings suggest that miransertib may represent a viable therapeutic option for symptom management in patients with Proteus syndrome. However, further studies are required to assess its long-term safety and efficacy.
Conclusion
Patients with Proteus syndrome experience clinical improvement due to selective inhibition of the PI3K/AKT pathway by miransertib, whose function is to block abnormally high AKT1 activity—a key mechanism driving uncontrolled growth and cellular proliferation in Proteus syndrome. Miransertib administration not only slowed lesion growth but also improved mobility and stabilized associated symptoms, including pain and pulmonary and skeletal complications, reinforcing the therapeutic potential of AKT inhibitors in this context.
However, given the results obtained and the limitations of our study, we propose conducting longer-term studies with larger participant cohorts to evaluate the sustained safety and efficacy of miransertib. We also suggest exploring combination therapies with miransertib to enhance efficacy and mitigate potential adverse effects.
Finally, investigating specific biomarkers that predict response to AKT inhibitor therapy would be important, enabling more personalized and effective treatment strategies for patients with Proteus syndrome.
